- Have Any Question?
- (+880) 017238012739
- enviroco@gmail.com
Hypochlorous acid
Hypochlorous acid’s chemical formula is (HCIO). It is often used as a safe disinfectant to treat water sources, irrigation systems, industrial wastewater (sludge control) and food preparation surfaces.
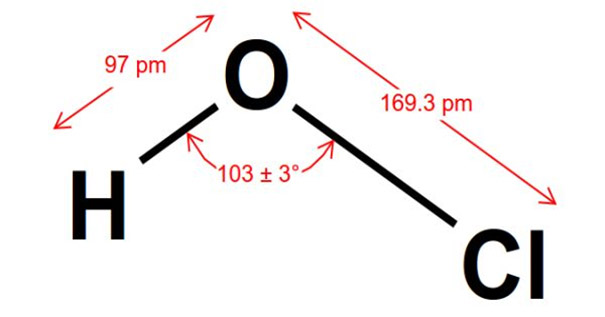
The hypochlorous acid with the name iupac is identified as monoxychloric acid (I). Having other different names, it is also called: chloric acid, chlorine hydroxide or hydrogen hypochlorite.
Hypochlorous acid has a partial dissociation in water because it is a weak acid and its acid dissociation constant (ka) is: 3.0 × 10-8.
In water, hypochlorous acid occurs both as a chemical chlorine solution and as water. It is a colorless solution and its exact physical properties are variable and they depend on the concentration of the solution.
Hypochlorous acid is a more potent disinfectant than chlorine, with antimicrobial and antibacterial properties against a wide range of microorganisms
General descriptions
Hypochlorous acid reacts with bases to form salts called hypochlorites. For example, sodium hypochlorite (NaOCl), the active ingredient in bleach, is formed as a result of the reaction of hypochlorous acid with sodium hydroxide.
The chlorine atom present in the hypochlorous acid molecule gives it a significant oxidizing power. In fact, its standard reduction potential is 1.61 volts.
The chemical formula of hypochlorous acid is HClO, the central oxygen is connected to the chlorine and hydrogen atoms by single bonds.
Hypochlorous acid was identified as a chemical entity more than 150 years ago. Its anti-infectious properties were already known before using aqueous chlorine as an antiseptic for traumatic wounds during the First World War.
In the 1940s, London hospitals used aerosolized solutions containing hypochlorous acid for the prevention of pathogens dispersed in the air.
Decades later, it was discovered that hypochlorous acid is produced in the human body by immune cells to fight infections because it works against a wide range of microorganisms.
Specifically, hypochlorous acid occurs naturally in activated human neutrophils and other tissue-dwelling phagocytes. This occurs by the activity of myeloperoxidase (MPO) on peroxides and on the cytoplasmatic during the “oxidative thrust" activated by the activation of phagocytes.
The hypochlorous acid generated physiologically is of short duration because the composite is highly reactive and is rapidly converted by oxidation and halogenation reactions. The antimicrobial effects on bacteria in phagosomes are fast and powerful. In contrast, the reaction products with intracellular proteins, amino acids and small molecules persist with much longer half-lives and participate in a series of downstream events.
Hypochlorous acid, Lewis.
The Lewis formula or Lewis structure is used to graphically represent the covalent bond of a molecule or ion. In Lewis’s representation, the symbols of the elements represent the atoms, while the points represent the electrons that surround them.
This method was invented by chemist Gilbert Lewis, based on the byte rule. This rule is empirical in nature and was formulated by Gilbert Newton Lewis himself to expose the chemical bond formations between atoms. This rule can only be used for the atoms of the main groups of the periodic table.
The byte rule says that “each atom tends, through the formation of chemical bonds, to acquire or lose or share the electrons to reach an external electronic configuration composed of eight electrons".
A Lewis structure can be designed for any covalently bound molecule, as well as for coordination compounds.
The Lewis structure for hypochlorous acid is shown below
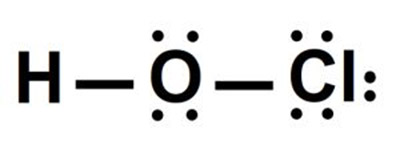
How is hypochlorous acid formed?
Hypochlorous acid can be produced in the laboratory by adding chlorine to water by obtaining hypochlorous acid with hydrochloric acid.
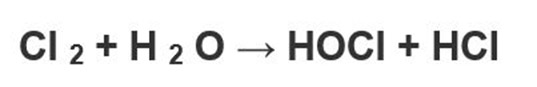
The above reaction is in equilibrium and it is not easy to isolate HClO from this mixture. However, stable hypochlorous salts can be obtained by dissolving chlorine gas in sodium hydroxide solution or other aqueous base solutions.
HClO can be prepared also by dissolving chlorine monoxide in water according to the following reaction.
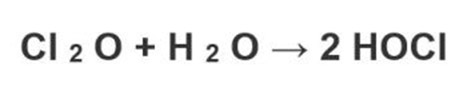
Another method for obtaining hypochlorous acid is to bubble CO2 into a suspension of lime chloride, Ca (ClO) 2. During the passage of CO2, it releases the hypochlorous ions derived from lime chloride that will be hydrolyzed in an aqueous solution according to the following reaction.
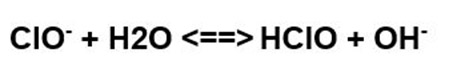
How does the hypochlorous acid dissociate?
Hypochlorous acid dissociates rapidly, especially in sunlight, developing oxygen and chloric acid. At first, dissociation with the development of oxygen is obtained in the form of the following reaction:

The oxygen generated in turn oxidizes the remaining hypochlorous acid according to the following reaction:
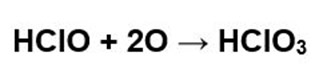
Hypochlorous acid, oxidation number.
The oxidation number of the hypochlorous acid is +1.
To identify the oxidation number of a component in a composite, the concept of electronegativity is used.
Using this principle, one can establish the oxidation number of an element. It is the value of charge that it assumes when one assigns the electrons of connection to the most electronegative element.
For hypochlorous acid (HClO), we have:
O = -2; H = +1; and therefore Cl = +1.
Hypochlorous acid, covalent bond (dative).
The chlorine present in hypochlorous acid has electronic doublets. In contrast, the chlorine of perchloric acid (HClO4) allows a covalent bond because it has doublets given to oxygen atoms. These atoms have six valence electrons that need two electrons to complete the valence layer and reach the byte.
Properties
Chemical formula: HCIO
Hypochlorous acid iupac name: Acide oxychlorique (I)
Hypochlorous acid ka, acid dissociation constant: 3,0 × 10-8.
Molar mass: 52,46 g / mol
Solubility in water: soluble
Liquid appearance: transparent.
Density: variable
CAS number: 7790-92-3.
ChemSpider: 22757.
EC number: 232-232-5.
UNII: 694C0EFT9Q.
RTECS number: 712K4CDC10.
ChEBI: 24757
PubChem CID: 24341.
Complete the following form and tell us about your project. We will contact you as soon as possible
